
Just two big deals, Bristol-Myers Squibb’s $74bn pending acquisition of Celgene and Abbvie’s $63bn outlay on Allergan are set to make 2019 a strong year for biopharma M&A, but there are plenty of other indicators of a buoyant market.
A new report from Vantage, part of the Evaluate group, says the first half of 2019 has also been a good year for medtech finances, thanks to the $1bn fundraising Google’s healthcare arm Verily achieved in January.
Vantage say these big deals have actually masked an overall decrease in the total number of M&A activity as well as a decline in licensing deals.
These trends have been dampened down by bullish stock markets, which have helped biotech raise $2bn in IPOs in the second quarter alone. The biggest biotech flotation in the first six months was the $348.5 IPO in June by BridgeBio, the San Francisco biotech focused on genetic diseases. The biotech has also managed to increase its share price since its launch, a feat not frequently achieved in the sector.
The second half of the year has begun with Denmark’s Genmab raising a huge $506 million via its debut on the US Nasdaq market in early July, which could signal a new surge in confidence in European biotech.
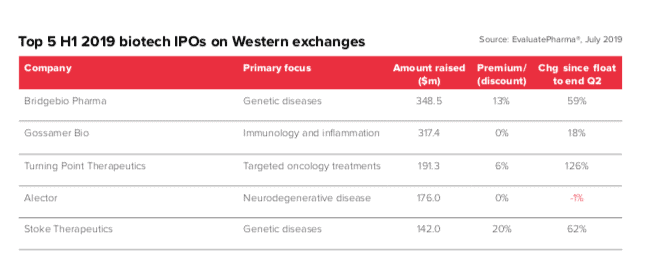
Venture capital is also fuelling biotech funding, with $3bn raised in Q2.
“We’re seeing strong interest in biopharma and medtech companies from public and private investors, though certain areas are benefitting more than others,” said Vantage report author Amy Brown.
“This likely affects the volume of acquisition and licensing deals. Barring a dramatic decline on the financial markets, it’s hard to see this trend shifting, although the potential for new drug pricing policy could make investors more jittery in the coming months.”
Other highlights from the period include the unexpectedly robust performance of Roche, which has shrugged off the attack of biosimilars in Europe to record a 9% increase in Q2 revenues, and a 13% increase in share price in the first half of 2019.
AbbVie and Bristol-Myers Squibb were the lowest performers in this segment, with doubts dogging their big merger deals, though both could see a resurgence once the deals go through and key drugs gain approval.
Another bellwether, the number of FDA approvals for novel drugs, is well down on last year. The US regulator has approved 18 innovative medicines in H1, with 2019’s total tally expected to be 45 – far below the 62 achieved in last year’s record breaking run.
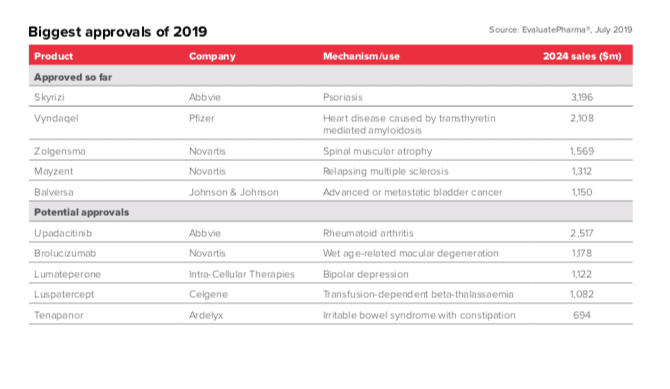
This decline isn’t worrying markets, the industry or regulators unduly, however, with FDA stalwart Janet Woodcock promising that the agency will maintain the reforming programme established by the energetic and now former FDA commissioner Scott Gottlieb.
Read the full report here: Vantage Pharma, Biotech & Medtech Half-Year Review 2019




