In Westminster, the Brexit uncertainty continues, but what is now beyond doubt is that the UK won’t be leaving the EU on 31 October, with or without a deal.
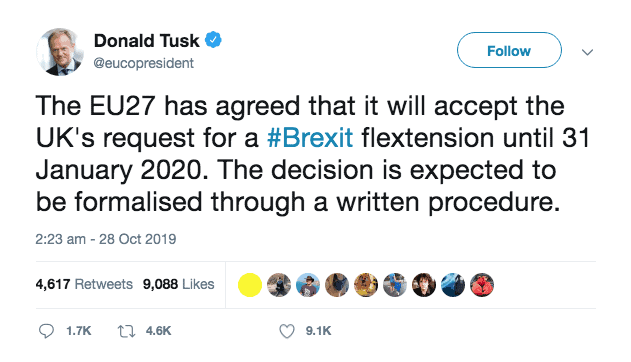
As it stands, the European Council has agreed to a Brexit delay until 31 January 2020, according to European Council president Donald Tusk.
EC president Donald Tusk confirms Brexit delay
It looks like EU27 leaders favoured a short technical extension, to allow Prime Minister Boris Johnson (pictured below, right) more time to win support for his withdrawal agreement, over a lengthier delay – three months or even a year – that might have given the UK time to unlock the impasse in the Commons.
What happens now?
The latter option allows time for a snap election – Johnson’s favoured option given the Conservative’s lead in the polls over Labour – or potentially even a confirmatory vote on whether Brexit should still go ahead at all.
Former PM Tony Blair has suggested a simultaneous general election and a second referendum, saying there is nothing to stop both being organised at the same time.
The EU met this morning to consider a Brexit extension until 31 January, with an option for the UK to leave earlier if a deal is ratified. This ‘flextension’, which will allow the UK to leave before the new deadline if a deal is approved by Parliament, has been agreed by the EU.
In a series of votes at the end of last week, Johnson’s revised withdrawal agreement was voted down by Parliament, then given support in principle, only to be effectively placed on hold by a subsequent rejection of his proposed three-day timetable for it to be debated in the house.
That meant that, despite vehemently opposing any extension, Johnson was forced to send an unsigned letter requesting more time in order to meet the requirements of a law passed by MPs – known as the Benn Act – with a second signed letter arguing against a long delay. The Benn Act orders the government to accept a delay if offered.
Election push
The government has tabled a motion asking MPs to debate and vote today on a general election for 12 December, the third time it has tried to convince the house to back one, in return for more time to scrutinise and debate the withdrawal bill.
In principle, if MPs were to agree an election – and also pass the withdrawal bill – Brexit could still take place before an election. Labour’s official position, however, is that it will block the move, which requires a two-thirds majority under the Fixed-term Parliaments Act (FTPA).
With more than a third of MPs, the main opposition party effectively has a veto, and is uncomfortable with an election given its terrible standing in the polls, not to mention its contradictory position on Brexit as a whole. The official line is Labour wants to defer any election until a no-deal Brexit has effectively been ruled out.
Johnson’s push for an election could be helped by smaller opposition parties – particularly the Liberal Democrats and Scottish National Party – who expect to do well at the ballot box. Once again, the proviso is that the withdrawal agreement bill is shelved and the threat of a no-deal Brexit removed.
They are backing a separate, one-line bill specifying that there will be an election on 9 December, which would require a simple majority.
That would, however, be open to amendments, for example, giving a vote to 16- and 17-year-olds or the estimated two million UK citizens living in the EU. Downing Street has been reluctant to go down that route until it no longer has any chance to get the current version of the withdrawal bill approved, although reports this morning suggest it may be warming to the idea.
 So once again, the path forward is as clear as mud, and industry remains bereft of the certainty it craves to allow effective forward planning and investment decisions.
So once again, the path forward is as clear as mud, and industry remains bereft of the certainty it craves to allow effective forward planning and investment decisions.
Meantime, Johnson has also threatened to obstruct Parliamentary business by repeatedly tabling the bill until it goes through or until he wins support for an election. It’s not clear if that would be permitted by House Speaker John Bercow (pictured above).
For pharma, the new version of the withdrawal agreement retains the transition period in Theresa May’s version of the deal in which medicines and devices regulations would stay aligned with the EU. That means, for example, companies could continue to carry out batch release testing in the UK and manufacturing and distribution licenses would continue to be recognised until the end of 2020.
There would no longer be UK participation in EU institutions like the EMA, and while the Medicines and Healthcare products Regulatory Agency (MHRA) would be excluded from the EU regulatory network it would still have access to EU databases. The future relationship would be negotiated as part of the Free Trade Agreement negotiations.
 Health Secretary Matt Hancock (pictured right) told the Bioscience Forum earlier this month that the intention is for regulatory collaboration with the EMA to continue, including mutual recognition of products produced in the UK and the EU, according to a BioIndustry Association (BIA) Brexit webcast last Friday.
Health Secretary Matt Hancock (pictured right) told the Bioscience Forum earlier this month that the intention is for regulatory collaboration with the EMA to continue, including mutual recognition of products produced in the UK and the EU, according to a BioIndustry Association (BIA) Brexit webcast last Friday.
A new Northern Ireland problem for pharma?
From a pharma perspective, the new withdrawal agreement – which aims to tackle the issue of the Northern Ireland backstop – raises an important consideration.
If no trade deal is agreed before the transition period comes to a close, medicines regulation in Northern Ireland would be aligned to the EU, which might mean the MHRA having to enforce EU legislation there but possibly divergent UK rules on the mainland, said BIA chief executive Steve Bates.
Meanwhile no-deal preparations continue, and it has been reported that the government has now secured additional ferry routes for use in the event of a no-deal, while the Department of Health has also procured rapid air routes for essential medicines.
The BIA advised last week that companies seeking to use that capacity in the event of a no-deal should check with the civil service contact they have registered with.