Executive summary
- The Japanese economy, the third largest in the world, has been struggling with stagnation for decades, made worse by the triple disaster (earthquake/tsunami/nuclear leak) of 2011
- Health indicators are excellent with the longest life expectancy and among the lowest infant mortality rates in the world, but an ageing population presents particular challenges for policy makers
- The disease profile is changing, with rising cancer and heart disease, as people live longer, modify their traditional diet and become more sedentary
- Japan spends 8.3 per cent of its GDP on healthcare, below the OECD average, achieving good quality care, but that is now at stake with doctors and patients demanding higher standards
- The March 2011 earthquake has highlighted structural problems in the healthcare system, which is facing demographic, economic and political pressures
- The Japanese pharmaceutical market was worth around U$96.5bn in 2010, according to IMS Health, and the second largest in the world after the US, but its growth rate is stagnating
- A ‘price premium’ was created to reward innovative drugs on a trial basis and some form of this is likely to be adopted after the biennal price review of April 2012
- The pharma industry body is campaigning to make the price premium permanent and speed up approval rates to fight what it calls an ‘unfavourable environment’ for the industry
- The market is likely to split in two: high-end drugs that qualify for top prices and generics
- The trend of Japanese companies investing in other Asian markets is set to continue on the back of a strong Yen and a globalisation strategy
- Generics offer great opportunities, as the government is backing them more vigorously and educating reluctant patients on the merits of unbranded versions to face the growing healthcare bill
- Investing in Japan can be advantageous for R&D companies because it is a mature, ‘safe’ market with relatively low competition from generics and OTC and low parallel imports. However, generics are growing and price cuts during patent periods eat into profits
- Japan is still strong on innovation and is actively trying to fight ‘drug lag’ and create a more hospitable environment for clinical trials and biotech start-ups
- Risk aversion and old business models are dampening the viability of biotech start-ups, despite government efforts and high quality biomedical research.
Introduction
| Overview Population: 127,450,459 |
Japan has the world’s third largest economy and its growth has been remarkable since World War II. In the 1990s, when the asset price bubble burst, the country entered what economists called the ‘lost decade’, which has turned into 20 years of stalled growth. Banking, public spending and private sector reforms were implemented with an emphasis on frugality and risk aversion.
However, changes have been slow and Japan remains a traditional, hierarchical society contending with the rise of its rival China and an increasingly ageing population.
The gloomy economic scenario of recent years, marred by political paralysis, darkened considerably with the devastating earthquake and subsequent tsunami of March 2011 in the northeastern coast, which caused a nuclear explosion in the Fukushima Daiichi atomic plant.
This was the worst calamity Japan has suffered in half a century, with a death toll nearing 20,000 people. Reconstruction, safety issues and relocation of the evacuees, combined with control of the country’s public debt (twice the size of the economy in 2011) are major challenges for the government. The energy infrastructure and industrial production (including pharmaceuticals) were disrupted, but there are signs of economic recovery despite the debt, deflation and the high yen. In order to stabilise financial markets, the Bank of Japan pumped more than $325bn into the economy. Estimates quoted in the Wall Street Journal suggest direct costs of the damage – rebuilding homes and factories – ranging from $200bn to $300bn. The government also lowered its growth forecast for 2012, saying the economy would grow by 2.2 per cent, down from its earlier target of as much as 2.9 per cent.
Economist Dan Ryan, from IHS Global Insight told PME: “After a sharp contraction, production gradually improved and unemployment, excluding the three hard-hit prefectures, remained moderate. Coupled with the gradual rebuilding effort, this could have been an opportunity for Japan to increase its GDP growth rate significantly. However, the global economy in late 2011 was sluggish. As a result, Japan’s exports, which depend on foreign demand, were weakening … Given that yen-denominated export prices have been weak, this confirms that the problem is falling demand, not supply constraints left over from the earthquake or from Thailand floods affecting Japanese assembly firms.”
| Top 10 causes of death | |
| 1 | Cerebrovascular disease |
| 2 | Influenza and pneumonia |
| 3 | Coronary heart disease |
| 4 | Lung cancer |
| 5 | Stomach cancer |
| 6 | Colorectal cancer |
| 7 | Liver cancer |
| 8 | Suicide |
| 9 | Kidney disease |
| 10 | Pancreatic cancer |
Last year’s disaster also produced another seismic change: Yoshihiko Noda from the Democratic Party became the sixth Prime Minister in five years in August 2011, following the resignation of Naoto Kan, who had been tarnished by the economic recession and the response to the nuclear crisis in the aftermath of the earthquake.
Noda is committed to fiscal reform, as Japan has the lowest tax rate in the Organisation for Economic Cooperation and Development (OECD), (17 per cent of GDP) giving room to raise taxes.
The conservative Liberal Democratic Party had been the dominant political party, holding power almost continuously for 54 years, until it was heavily defeated in 2009.
Japan is a constitutional monarchy where the Emperor (currently Akihito) is merely a ceremonial figurehead. Executive power is held by the Prime Minister and the legislative organ is the National Diet, a bicameral Parliament.
Healthcare
Japan spends only 8.3 per cent of its total GDP on health according to the World Bank and of that, more than 80 per cent is public expenditure that has been decreasing in real terms, as debt reduction is the priority.
Extremely positive health outcomes have been attributed to universal coverage, lifestyle, effective government campaigns and high literacy. In the mid-1990s the pace of decreasing mortality, particularly in adult males, has been much slower, compared with other developed nations. Explanations offered by medical experts refer to high tobacco consumption (almost 39 per cent in males), changes in diet and rising suicide rates, especially following the earthquake/tsunami/nuclear disaster in 2011.
The number of suicides in Japan has stayed at more than 30,000 a year (24 for every 100,000 people) for more than a decade, despite a series of measures to try to reduce this figure, involving local governments, non-profit organisations and medical professionals.
The number of suicides rose after the Kobe earthquake in 1995, which killed nearly 6,500 people and the British Medical Journal reported in September 2011 that ‘Japan’s suicide rate is expected to rise after triple disasters in March’. Management of chronic illnesses and mental health problems such as post-traumatic stress among evacuees was also critical after the disaster.
Japan’s disease profile is changing, with a higher incidence of cancer, although survival rates for breast, cervical and colorectal cancer are among the highest in the OECD despite low screening rates for breast cancer, for instance, as there is no national programme. In 2007 only 24 per cent of eligible women were screened for breast cancer, compared to an OECD average of 62 per cent.
Ministry of Health, Labour and Welfare (MHLW) data revealed that the largest single cause of death was cancer, followed by stroke and heart disease. The latest available data shows that the top-selling therapeutic drugs in Japan continue to be those for cardiovascular disorders.
The Yano Research Institute of Tokyo published a 10-year forecast in 2009 saying that patient numbers would increase in the following categories: cancer, cardiovascular, mental disorders/depression, menopause and allergies. It also predicted that oncology products would drive the market.
Only about 3 per cent of the Japanese are obese, compared with more than 30 per cent of Americans, according to OECD data, but the numbers of sedentary, overweight Japanese are growing and there is now mandatory screening for the majority of the population and counselling for weight problems.
“The Japanese health system that had worked in the past has begun to fail, and is now threatening human security within Japan, as exemplified by the recent disaster … The financial and social underpinnings of healthcare in Japan are now under threat from economic stagnation, which has widened social divisions. As inequalities increase, social determinants of health will likely dete-riorate, leading to poorer population health and greater demand for services,’ argues The Lancet, which recommends a structural reform of Japanese healthcare, suggesting the country could become a global leader by sharing its scientific knowledge more efficiently. (‘Japan: Universal healthcare at 50’, August 2011).
Kaih oken – universal cover
The first Employee Health Insurance Plan, known as ‘kaihoken’, was created in 1927 and expanded in 1961, with costs starting to increase in the 1980s.
Kaihoken means that healthcare in Japan is free and for all. It is a universal system based on compulsory insurance plans that covers employees (under 70 years old, the so-called ‘pre-elders’) and their families, with patients paying around 30 per cent of medical costs. Those without company coverage, such as students or selfemployed professionals, can access a national health insurance programme provided by local government and the poor receive free care. The segment of the population over 70 years old pays only 10 per cent of check-ups, treatment and medicines.
Within the National Health Insurance (NHI) scheme, patients enjoy total freedom to choose doctors, specialists and to decide where they receive treatment. Waiting lists are virtually non-existent. Most of the hospitals are run by medical practitioners as private institutions and a small percentage are run as non-profit organisations.
Every two years, prices for medical services and products, such as pharmaceuticals, are set by a government committee after negotiations between the Health Ministry and industry providers and a huge list of fees for treatments/medicines is published (the Yakka Kijun, or reimbursement tariff list). Until recently, no adjustments or exceptions were permitted and the government maintains a tight grip on health finances. Although lower in price than in other industrialised nations, services are used intensively.
Compared to other countries, the Japanese visit the doctor more often and hospital stays are, on average, longer. There are no gatekeepers and patient satisfaction with choice is high. However, doctors complain that they are not paid enough and have to find ways to supplement their income, particularly in the case of highly specialised consultants, who may also run pharmacies. According to The Economist, doctors tend to overprescribe tests and drugs and keep patients in hospital (‘Not all smiles: Japan’s healthcare system is the envy of the world. It is also in crisis’, September 2011).
Consultations with doctors
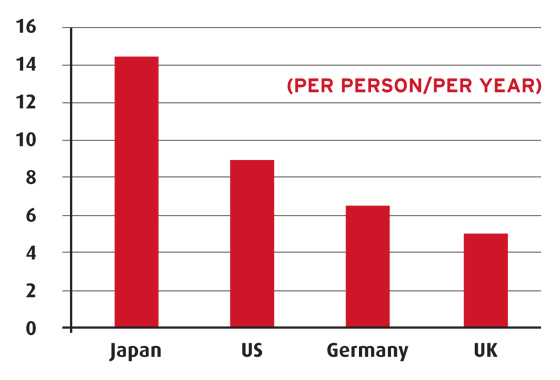
Source: OECD, most recent by country
Japanese doctors tend to prescribe relatively weaker doses of drugs compared to other countries, both because of physiological differences and as a result of high pharmaceutical consumption (because physicians profit from dispensing drugs), according to the OECD.
Supplementary private health insurance is available to cover the co-payments or non-covered costs, but even dental care is part of the NHI package.
Overstretched system
In the last few decades, Japanese policy makers have started to focus on how to address the issue of an ageing population.
However, instead of allocating more funds, the government has lowered fees for consultations and medical tests. Patients favour medical technology, such as scans, having them eight times more than British patients, for instance.
The system is stretched to the limit and open to over-medication and over-testing with low out-of-pocket expenses incurred by patients. However, surgery rates are low as, culturally, operations are considered invasive procedures and there is no financial incentive for surgeons, since they are paid fixed salaries.
Emergency care services are inundated with patients seeking routine treatments, and chronic disease management and preventative care are not so good. There is also a GP shortage in some areas, particularly in the cities, as salaries are higher in rural areas.
There are shortages of obstetricians, paediatricians, anaesthetists and emergency specialists, owing to state quotas, relatively low pay and long hours.
Hospital beds per 1,000 people
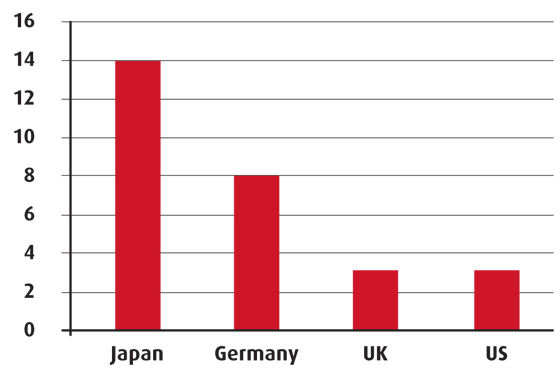
Source: World Bank, 2008/2009
In the long term, this almost ‘perfect’ system may well be unsustainable, as Japan’s disease profile changes and the population continues to age, with a diminishing young workforce to pay the bills. By 2050, 40 per cent will be 65 or older and the fertility rate, at 1.2, is one of the lowest in the world.
Costly diseases, such as cancer, stroke and Alzheimer’s disease are becoming more prevalent, while the emphasis up to now has been on primary care and prevention of, for example, obesity.
Patients have grown to expect better and more targeted services, as well as accountability, but this has not been reflected by the medical profession, which has focused on offering above standard primary care but without taking into account social changes and rising expectations.
A 2010 opinion poll by Japanese experts cited in The Lancet suggests that the major reasons why patients are dissatisfied with the health system are not to do with quality, choice, access, or costs, but rather the lack of fairness and public participation in decision making and these concerns have increased in recent years.
The accreditation system for medical subspecialties is not well established, which means doctors can claim and practice any speciality they desire. Furthermore, national quotas for training subspecialists based on the expected need, and the resources for meeting the required level of experience, do not exist. General Practice is yet to be recognised as a speciality.
Pharmaceutical market
The Japanese pharmaceutical market is the second largest in the world, after the US, worth around U$96.5bn in 2010, according to IMS Health. CAGR (in $ at constant exchange rates) was 2.6 per cent between 2006–2010, and CAGR of 2–5 per cent is projected between 2011 and 2015.
Recent growth rates have been significantly inflated by currency fluctuations between the Japanese Yen and US Dollar.
The largest therapeutic category is Renin-angiotensin system agents (antihypertensive), followed by anti-neoplastics (cancer drugs).
Market share of global pharma sales
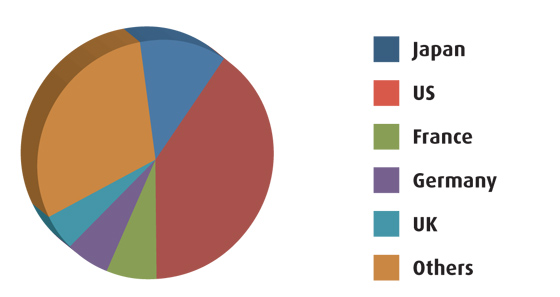
Source JPMA citing IMS Health data for 2008
Pharmaceutical businesses were disrupted by the earthquake disaster, which forced manufacturing plants to stand idle or struggle at partial capacity. This added pressure to the Japanese pharma industry, which was already struggling with a stagnating economy, increasing competition from China and India and regular price cuts. In fact, Espicom projects that Japan will drop to become the second largest pharmaceutical market in the Asia-Pacific region, likely to be overtaken by China in 2016.
In recent years Japan, like other markets, has been affected by the patent cliff. Analysts forecast a continued flurry of mergers and acquisitions, as investment dwindles.
Ian Haydock, Asia editor at Scrip told PME: “Market growth will be dampened by the biennial price cut in April, while top company Takeda is expecting the first US generic competition to its multi-billion dollar main product, the antidiabetic Actos (pioglitazone), in August. Previous price revisions and ongoing discussions suggest that April could see a mixed bag of changes for manufacturers, with a tweaking of Japan’s complex price calculation system to provide further incentives for new drugs being offset by price cuts for older drugs, for example.”
The bulk of raw materials is imported and many Japanese companies are choosing to conduct human clinical trials in Australia, where the regulatory environment is more favourable and it is quicker than in the US, for instance.
The pharmaceutical market is highly fragmented, with over 1,000 companies, hundreds of which are very small enterprises selling traditional Chinese medicines.
Up until the 1990s, the environment favoured the local pharma industry, but market liberalisation has since brought stiff competition from foreign firms and takeovers of Japanese companies by multinationals.
The internationalisation of the Japanese pharmaceutical industry has advanced rapidly in recent years and large companies are endeavouring to extend their product pipelines through acquisitions or alliances with biotech companies.
The approval of Halaven (eribulin, Eisai) for progressive breast cancer in the US in late 2010, and in Europe and Japan in early 2011, signalled the impact of a growing number of agents that Japanese pharmaceutical companies are introducing to the global oncology markets.
In 2007 the government established a Five-year Action Plan for the Creation of Innovative New Drugs and New Medical Devices to deal with issues such as drug lag. The size of the Japanese pharmaceutical market, which about 10 years ago was approximately 20 per cent of the global market, has now shrunk to just 10 per cent.
Best-selling drugs in Japan
| Drug (brand) | Generic name | Company | Category | |
|---|---|---|---|---|
| 1 | Aricept | donepezil | Eisai | Alzheimer’s disease |
| 2 | Blopress | candesartan | Takeda | Hypertension |
| 3 | Diovan | valsartan | Novartis | Hypertension |
| 4 | Cervarix | human papillomavirus (HPV) vaccine | GlaxoSmithKline | HPV |
| 5 | Lipitor | atorvastatin | Pfizer | Cholesterol lowerer |
IMS Health Japan (July to September 2011, Q3)
The international appeal of Japan’s drug discovery environment and medical drug market has also fallen, according to the Japanese Pharmaceutical Manufacturers’ Association (JPMA). The industry body considers that the primary causes behind the current crisis are: ‘Japan’s drug price system, delays and increased costs for clinical trials, and delays in the new drug approval process’.
The JPMA is a member of the Federation of Pharmaceutical Manufacturers’ Associations of Japan (FPMAJ) that comprises several industry bodies, such as the generics association, OTC manufacturers, self-medication and also regional bodies focusing on domestic issues. The JPMA has a more global reach, but drug lag and tight price controls are common ground and both organisations engage in joint campaigning.
Pricing and reimbursement
Pharmaceutical expenditure accounts for 1.8 per cent of the country’s GDP. To control expenditure further, the government has lowered prescription drug prices and tried to separate prescription and distribution.
For drugs and devices, the price is revised biennially, mainly based on the results of a market price survey. Reimbursement levels for drugs are recommended by the Central Social Insurance Medical Council (Chuikyo), a consultative group consisting of representatives of the government, the public, the medical profession and various other groups.
Price cut comparison
| Number of products subject to price cuts in 2008 | 12,740 |
| Number of products subject to price cuts in 1992 | 2,121 |
Source: JPMA 2011
The prices of new drugs are set by comparing their efficacy and innovativeness with existing ones. Additionally, if sales of a new drug are greater than estimated by the manufacturer, then its price is unilaterally lowered by the MHLW.
However, companies can claim a tax credit of 8 to 10 per cent for R&D on the total cost base. Almost all approved drugs are reimbursable at some level, apart from oral contraceptives, lifestyle drugs and some vaccines. Complex reimbursement formulae account for dispensing fees, counselling and administration, cots and medication supplies and devices. Additional adjustments are made based on dosage form, length of therapy, number of prescriptions dispensed by the pharmacy per month, and when the prescription is filled.
The regulatory agency is the Pharmaceuticals and Medical Devices Agency (PMDA), formerly the Kiko Advisory Board, which conducts scientific reviews of marketing authorisation applications of pharmaceuticals and medical devices and monitors post-marketing safety.
The JPMA proposed a new premium price for drugs during patent periods for which generics have not entered the market, based on innovation, comparision of effectiveness with existing drugs, paediatric use, market size and other criteria. This pricing system started on a trial basis in 2010 and the industry wants it adopted permanently in 2012. Increases in healthcare spending would then be offset with a form of generic reference pricing on older, longlisted products. This pricing mechanism takes into account clinical benefits, allowing higher prices for truly innovative products.
Trial premium pricing scheme (operated since 2010)
| 70-120% | Innovativeness |
| 35-60% | Usefulness I |
| 5-30% | Usefulness II |
| 5-20% | Paediatric care |
| 10-20% | Marketability I orphan drugs |
| 5% | Marketability II small market scale |
Source: JPMA 2011
In 2010, of 15,455 products reviewed, 624 were categorised innovative, 12 were paediatric or orphan drugs and four were considered to have proven effectiveness, allowing for prices above the recommended threshold.
The FPMAJ is concerned that the 2012 price review may not include full implementation of the premium price allowance for new drugs and a price freeze for drugs for high medical needs, effectively a moratorium period. Pricing reforms are inevitable but they will be very unpopular. Companies are already reluctant to launch new therapies because of price cuts and drug lag, even with government efforts to make Japan more competitive.
Back to top of page
Japanese globalisation
Takeda remains Japan’s largest national pharma company, followed by Astellas, Daiichi Sankyo, Eisai and Shugai. Takeda’s patent on the blockbuster Actos (pioglitazone) for type 2 diabetes expired in January, but an agreement with generic drugmakers Ranbaxy, Watson and Mylan delayed its marketing until August 2012. Sandoz, Teva Pharmaceutical Industries and a few others will launch their own versions later. Actos accounts for around 27 per cent of Takeda’s annual revenue, a considerable impact on its profit.
Takeda also faces lawsuits involving alleged side effects of Actos. Last year, the US Food and Drug Administration reported that longterm Actos users were at a slightly increased risk of developing bladder cancer. Actos was withdrawn from the market in Germany and France, but not in the US.
Takeda’s strategy has been to diversify and extend its global appeal. It launched a new division in January 2012 to expand its vaccines business globally, testing a new vaccine to prevent Haemophilus influenzae type b and developing a human papillomavirus vaccine and a flu vaccine. It received government subsidies to focus more on vaccines, as the government’s strategy is to develop domestic production.
Takeda acquired Swiss company Nycomed in 2011 to increase its presence in Europe and gain access to emerging markets where Nycomed has a strong footing. The company is cutting 2,800 jobs, or about 9 per cent of its workforce, over the next five years, mostly in Europe, where subsidiaries will be merged or closed.
In 2008, Takeda bought US company Millennium Pharmaceuticals, the largest foreign acquisition ever by a Japanese company.
Leading companies (sales)
| 1 | Takeda Yakuhin Kogyo |
| 2 | Pfizer |
| 3 | Daiichi Sankyo |
| 4 | Mitsubishi Tanabe |
| 5 | Chugai Seiyaku |
| 6 | Novartis Pharma |
| 7 | Eisai |
| 8 | MSD |
| 9 | Astellas Seiyaku |
| 10 | Sanofi |
Source IMS Health Japan 2010
Rival Daiichi Sankyo, which acquired Indian generic firm Ranbaxy in 2008, also bought California-based Plexxicon to strengthen its oncology portfolio.
Since the 1980s, the Japanese pharmaceutical industry has been characterised by intense research and innovation as companies lost protection from international competition. Prices have also lowered significantly. Japanese firms have had to find profits elsewhere, leading to increased R&D budgets and the establishment of research centres abroad to tap into local knowledge.
According to IMS Health, Japanese companies controlled 85 per cent of the market in 1990. By 2003, Pfizer, Roche, Novartis and Merck had grown their presence, securing places in the top 10. Pfizer also dislodged Takeda from the top position in terms of market share.
Generics
The government set a target of 30 per cent market share by volume for generics by 2012 and has increased efforts to encourage generic prescription and substitution, which has angered doctors, though they retain the ‘right to prescribe’ what medicines they choose. Generics now hold a 25 per cent market share and most products are manufactured domestically. The challenge in Japan is that cheapness is associated with lower quality and brand trust is very important in a country where patients are suspicious of foreign drugs, particularly those manufactured in other Asian countries.
Nor is the price differential between branded products and generics significant (about 70 per cent of the original price). A survey by Chuikyo in 2007 indicated that doctors, revered as figures of authority, rely on patients to ask for generics, which only happens in 10 per cent of cases. Conformity and respect for authority mean that patients trust doctors and do not question their choices. Doctors also lack education regarding generic names of drugs.
Pharmacists are required to provide information about bioequivalence to patients at the point of substitution, which is timeconsuming, although they receive a modest incentive fee. The regulatory process is cumbersome and generic manufacturers must supply all strengths and dosage forms of the branded products. R&D firms aggressively protect patents, and to navigate the system most international generic companies partner with domestic firms.
The scope for growth is significant, with the government’s impending reforms to meet the need for drugs at affordable prices, even with increases in patient co-payment.
Foreign companies have shown interest in this untapped potential. Israeli giant Teva entered an agreement with local company Kowa creating a joint venture that put it among the top five generic players in the market, then acquired 57 per cent of Taiyo, Japan’s third-largest generic drugmaker and then bid for the rest.
Ian Haydock commented: ”The generics sector continues to make steady gains, driven by the loss of expiry for several big-selling drugs, increasing public acceptance and the expansion of major players like Teva.”
OTC drugs are divided into three categories depending on side effects and a pharmacist must always be present to dispense and give advice. Vitamins, supplements, energy drinks and cough drops are sold freely in supermarkets and other shops. Prices are similar to those of European products and more expensive than in the US. Under the revised Pharmaceutical Affairs Act, supermarkets and other stores can sell non-prescription drugs, including cold tablets and aspirin.
Biotech
According to the Japanese External Trade Organisation, JETRO, the biotech sector in Japan was estimated to be worth around $309bn in 2010. However it remains less prominent due to some isolation and lack of R&D investment, ranked eleventh in the world by the Scientific American Worldview Report and Scorecard.
A survey by the British Royal Society places Japan in third place for the publication of science papers, which shows that despite its excellent reputation in science research, Japan seems to have lagged behind in the commercialisation of that research.
The first biotech boom occurred in the 1980s with DNA recombinant technology, when companies such as Kirin and Takara joined, followed around the year 2000 with an explosion of start-ups.
After that, the government defined specific strategy guidelines and implemented national reform of the university system, allocating more funds to the sector, as well as establishing regional biotech clusters. Politicians intended to address the problems of an ageing population and depletion of natural resources by investing in biotherapies and food biotechnology. Nevertheless the market was dominated by big pharma with old industrial practices and the characteristics of the Japanese financial system meant that there was little risk money for start-ups. A mini bubble in 2004 left many feeling uneasy about biotech.
Both venture capital investment as a percentage of GDP and the share of high technology sectors in total venture capital are extremely low in Japan compared with most OECD countries, says the organisation. Some researchers also suggest that the cluster plan adopted, replicating the San Francisco or Cambridge models in the US and UK, respectively, may not work as well in Japan, where business and industrial practices are different. Again, the earthquake and tsunami disaster have impacted what little progress was made in the last decade and the economic crisis and China’s growth had already dampened interest in Japan’s prospects.
Trends and Outlook
Even with the devastating aftershocks of the 2011 earthquake, Japan managed to resume economic production and the pharmaceutical industry showed it has been built on a solid foundation.
At 50 years old, the universal healthcare system is showing signs of strain in a country with the longest life expectancy in the world and other positive health outcomes. The disaster has exposed structural failures and fragmentation, as well as a lack of transparency and communication between government agencies.
An ageing population presents many challenges to policy makers and free coverage of expensive treatments is not sustainable.
Prevention campaigns, increases in co-payments and a boost in generics are likely to cut healthcare public expenditure. The government has a pro-generics stance that will help domestic and foreign companies grow, as patents expire.
“There are also positive trends elsewhere, with continuing initiatives to encourage the development of high-need drugs, and a shortening in approval times, which is leading to the approval of important innovative drugs and growth for those that market them,” concludes Ian Haydock.
The price premium for innovation is expected to be adopted to improve R&D budgets, strengthening Japanese industry interests but also signalling to multinationals that investing is Japan is still worthwhile. The trend of Japanese companies investing in other Asia markets is set to continue due to the strength of the Yen and the strategic plans of the Japanese government for companies to compete with China and India.

The Author
Catarina Féria Walsh is a freelance journalist specialising in the pharmaceutical industry.





